Details
Sodium hypochlorite is most often encountered as a pale greenish-yellow dilute solution referred to as liquid bleach, which is widely used as a disinfectant or a bleaching agent.
Sodium Hypochlorite Generator system
Sodium hypochlorite solution can be produced via electrolysis of brine water that is used to substitute highly-toxic liquid chlorine and chlorine dioxide disinfection system and hence is widely applied in large and medium-sized water plants.
Available chlorine 0.6-0.8% (6000~8000ppm)
Salt consumption: 3.5kg/kg available chlorine
Direct power consumption: 4.2kW/kg available chlorine
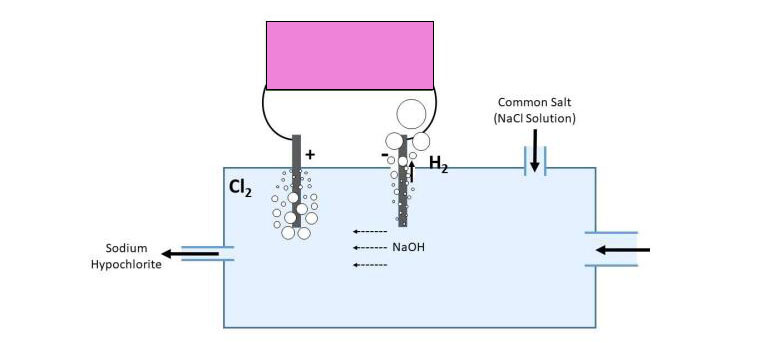
Reactions at the electrode are:
2Cl- → Cl2 + 2e- (positive electrode)
2H+ + 2e- → H2 , OH- + Na+ → NaOH (negative electrode)
NaOH produced near the negative electrode is carried away by moving water towards the positive electrode, where it reacts with chlorine to yield sodium hypochlorite
2NaOH + Cl2 → NaCl + NaClO + H2O
Application
Tap water plant disinfection, disinfection of municipal sewage, food processing, oil will disinfect water, hospitals, power plant cooling water disinfection.

